HFNC Oxygen Therapy For Adult Ventilators Replacement
In the spring of 2020, a plague swept the world, and tens of thousands of medical staff rushed to fight the fierce and cunning new crown virus on the front line of the fight against the epidemic, guarding human health.
When everyone is paying close attention to this epidemic, many soldiers have also received attention, and the high-flow respiratory humidification treatment device is one of them.
Some days ago, some friends who had never worked in the ICU asked, what kind of oxygen device the infected Wuhan doctor had on his nose, I have never seen it!
Today, I will introduce to you this high-flow respiratory humidification treatment device, a new type of respiratory support device that has emerged in recent years.
In clinical practice, we often see traditional oxygen therapy methods, which are mainly implemented through devices such as nasal catheters, nasal congestion, ordinary masks or oxygen storage masks. But these devices have two common disadvantages:
The first is that the oxygen flow rate they can provide does not exceed 15L/min. For some patients with acute respiratory failure, this cannot meet their needs;
The second is that the oxygen concentration provided by these devices is not accurate, and the oxygen concentration reaching the alveoli is usually reduced.
The high-flow respiratory humidification therapy instrument (HFNC) breaks through the shortcomings of traditional oxygen therapy. It can not only provide heated and humidified oxygen with a flow rate of up to 60L/min, but also accurately adjust the oxygen concentration.
The high-flow respiratory humidification treatment instrument is a new oxygen therapy method. The device is composed of an air-oxygen mixer, an active humidifier, a single heating circuit, a large-diameter nasal congestion catheter or a tracheostomy connector, and a mask adapter.
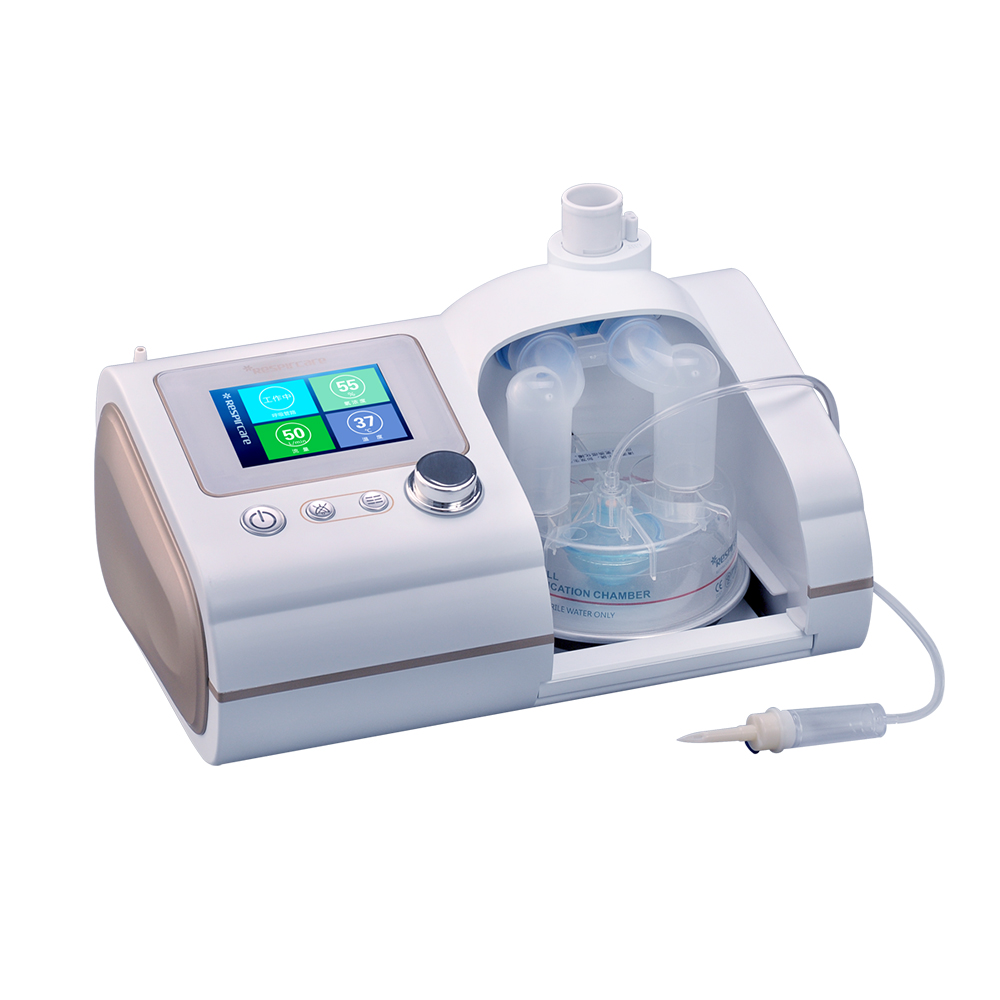
Main Features:
Delivering high flows , high concentration ,close to 100% relative humidity of air/oxygen mixtures to spontaneously breathing patients, is considered to have a number of following effects:
1. Keep constant、precise、high FiO2 and improve the alveolar ventilation
2. Improve the efficiency of breathing by reduction of anatomical dead space and replacing it with oxygen-enriched gas in the upper airway.
3. Low levels of PEEP may contribute to collapsed alveolar recruitment
4. Good humidity promote effective from the sputum and mucociliary clearance to reduce the risk of infection
5. More comfort , Better tolerance and Greater respiratory compliance in the patients.
Clinical application value
1. ICU emergency mild and moderate acute respiratory failure(hypoxic Non-CO2 retention )
Improve breathing difficulty and the effect of improving oxygenation is obvious.
The rate of intubation and mortality were significantly lower than that of noninvasive ventilation and standard oxygen therapy.
2. A new option from the creative withdrawal machine.
The drone fights and reduces the reintubation rate.
3. Acute heart failure or cardiogenic pulmonary edema
Improve oxygenation and reduce cardiac load.
4. Hypoxic patients assist bronchoscopy, transesophageal ultrasonography and digestive endoscopy.
Added an effective oxygen therapy.
5. Chronic respiratory failure
Improve ciliary activity, promote sputum discharge, reduce the chance of infection.
6. Palliative treatment
New options for end-stage patients who reject intubation and resuscitation.
7. Cardiothoracic surgery is associated with a risk of respiratory failure.
The results were comparable to non-invasive ventilation, but the patient was comfortable.
Model
| HUMID-BM
|
Power supply
| 220VAC+22V,50+1Hz
|
Flow setting range
| ●Low Flow Mode:2-25L /Min temp:34℃
●High Flow Mode:10-80L/Min
|
Temperature Control
| 31℃ ,34℃ ,37°C
|
Oxygen% Measure
| 21 %-100%
|
FiO2 Control
| Manual titration control
|
Alarm parameters
| ●Low oxygen concentration alarm
●High oxygen concentration alarm
●Ambient temp alarm
●Choke alarm
●Alarm recovery
|
| Recommended Ambient Temp | 18-28°C
|
Standards
| IEC/EN 6060-1, IEC/ EN 60601-1-2; IS08185
|
Package
| L330xW245xH315mm
|
Application
| HOSPITAL
|
Our Company
Shenyang Maisi Medical Technology Co., Ltd. is a high-tech emerging modern enterprise specializing in the development, production, sales and service of respiratory diagnosis and treatment and electronic products. It is a domestic company with core technology covering 2 major respiratory therapy equipment.
The company is headquartered in Hunnan High-tech Development Zone, Shenyang City, Liaoning Province. The company covers an area of more than 6000 square meters. The current main products are high-flow respiratory humidification therapy equipment (HFNC), non-invasive ventilation therapy equipment (BiPAP AutoBiPAP and Auto-CPAP) . The company's products have passed the US FDA, EU CE, RoHS and South Korea GMP certification, and strictly implement ISO13485, ISO9001 and national and industry related standards in every link of development and production. The product quality is stable and reliable, and has been universally recognized by customers all over the world.
The company's technical core backbone is composed of a team of technical experts from the fields of domestic information technology, automation control, machinery manufacturing and medical technology. The company has good cooperation with Air Force Aviation Medical Research Institute, China Medical University and other well-known domestic units.
Since its founding, Maxx Medical has forged ahead and has become a brand influential enterprise in the field of respiratory support in China. To create the future and not forget the original intention, Max Medical will strive to become an excellent brand of respiratory support in China.

Maisi has many years of experience in product development in the respiratory therapy industry, and has won a number of patents. The perfect after-sales service has brought us more and more users and sold to more than a dozen countries and regions. Based on the principle of good quality and customer first, we will dedicate effective and reliable products to customers with superb and innovative technical strength, advanced equipment and good service attitude.

Use advanced technology to serve clinical respiratory medicine and seek welfare for patients with respiratory diseases.
With professional integrity, we provide advanced, convenient and economical sleep and breathing treatment programs for patients with respiratory diseases.

FAQ
1. Are you a manufacturer or trading company?
RE:We are a manufacturer specilized in respirtory products,including Noninvasive Ventilators, HFNC and Oxygen Concentrators.
2. Do you have any certification?
RE:We are CE and EN ISO 13485 approved and FDA is in application and will be available in near future.
3. How long is your product warranty period?
RE:2 years.
